Vinylic Proton Nmr Shift
1 h nmr chemical shifts.
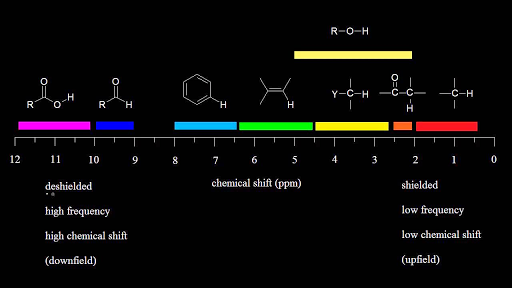
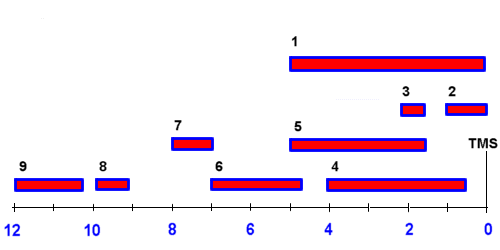
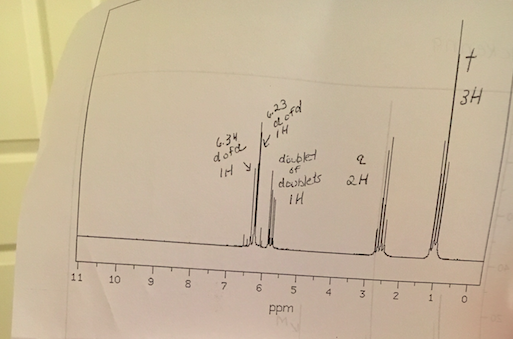
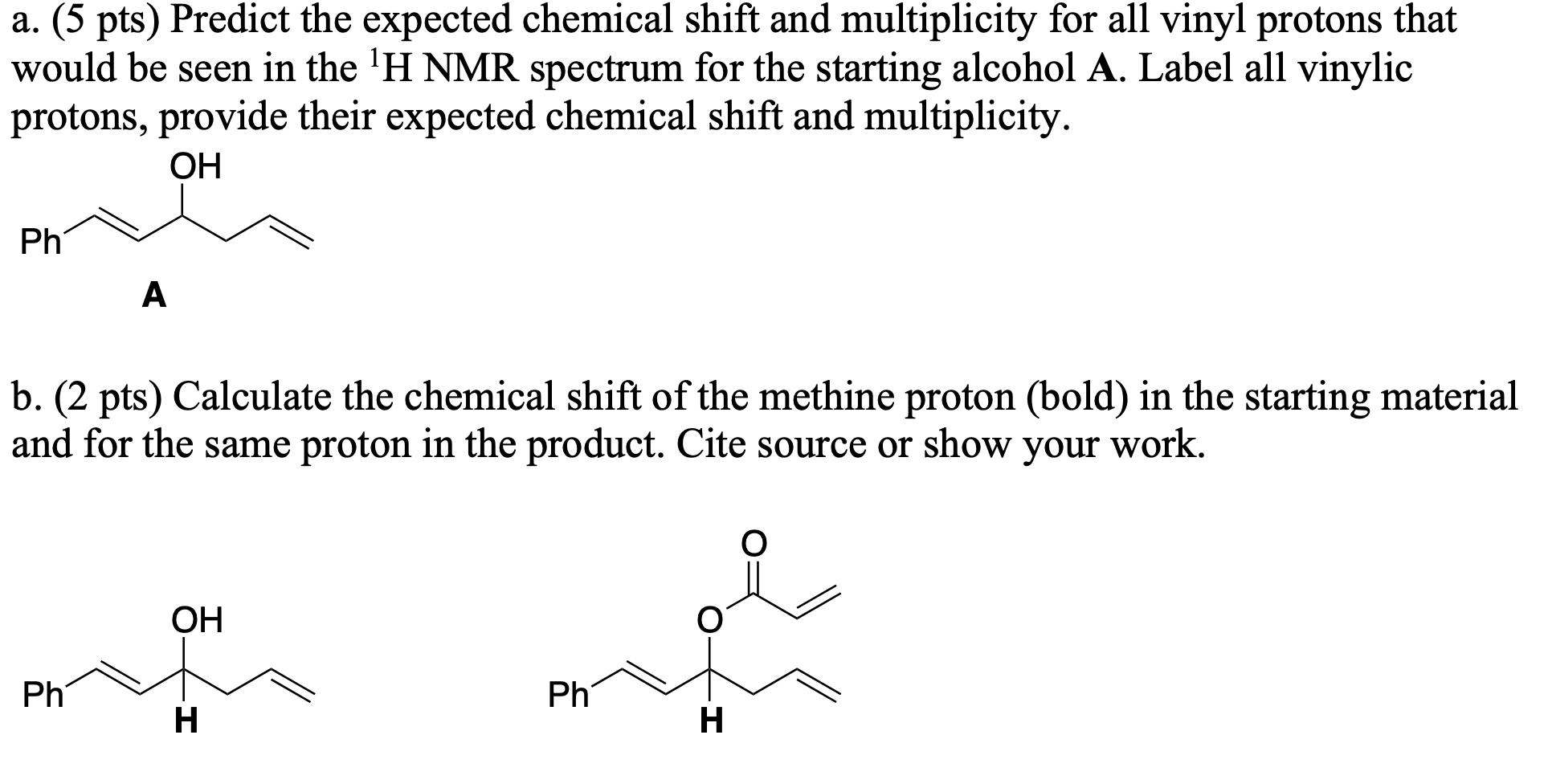
Vinylic proton nmr shift. The greater the substitution on the carbon bearing the hydrogen the further downfield higher frequency the resonance occurs. 30 c using 1h and 31p nuclear magnetic resonance spectroscopy. Table of characteristic proton nmr chemical shifts. Type of proton type of compound chemical shift range ppm rc h 3 1 aliphatic 0 9 r 2 c h 2 2 aliphatic 1 3 r 3 c h 3 aliphatic 1 5 c c h vinylic 4 6 5 9 c c h vinylic conjugated 5 5 7 5 c.
A summary table of chemical shift information is given in appendix iii. We know that a proton alpha to a carbonyl group is pulled downfield. Nmr chemical shift values table in the previous post we talked about the principles behind the chemical shift addressing questions like how the ppm values are calculated why they are independent of the magnetic field strength and what is the benefit of using a more powerful instrument. These methods which range from very simple to somewhat sophisticated are complimentary to one.
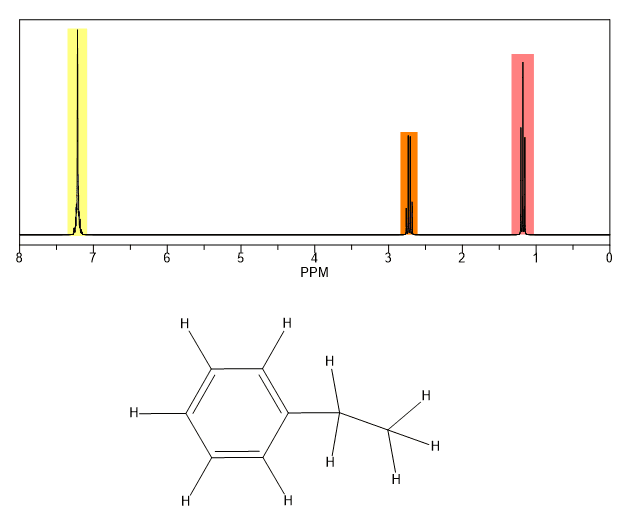
In other words frequencies for chemicals are measured for a 1 h or 13 c nucleus of a sample from the 1 h or 13 c resonance of tms. Table of characteristic proton nmr shifts type of proton type of compound chemical shift range ppm rch 3 1 aliphatic 0 9 r 2 ch 2 2 aliphatic 1 3 r 3 ch 3 aliphatic 1 5 c c h vinylic 4 6 5 9 c c h vinylic conjugated 5 5 7 5 c c h acetylenic 2 3 ar h aromatic 6 8 5 ar c h benzylic 2 2 3 c c ch 3 allylic 1 7 hc f. Characteristic proton chemical shiftstype of protonstructurechemical shift ppmcyclopropanec3h60 2primaryr ch30 9secondaryr2 ch21 3tertiaryr3 c h1 5vinylicc c h4 6 5 9acetylenictriple. Assigning the 1h nmr signals of aromatic ring 1h atoms assigning 1h nmr signals of 1h atoms on an aromatic ring based upon their chemical shift and coupling can be accomplished in a number of different ways which will be detailed below.
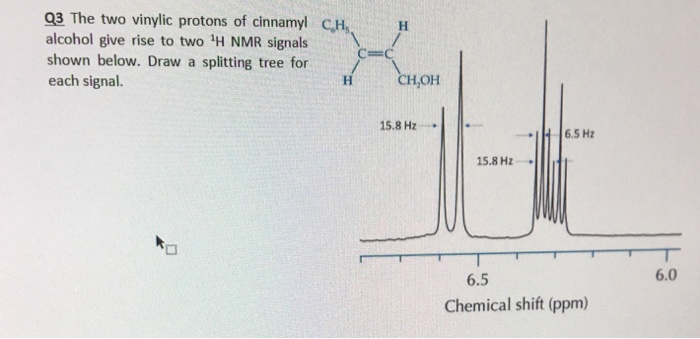
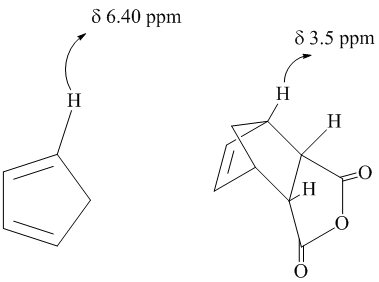
0 8 1 5 ppm alkane c h. Chemical shift d type of proton examples chemical shift in ppm comments. This is not surprising since the proton is not only vinylic but it is also alpha to a carbonyl group. Meo2c ch c nme2 co2me has a chemical shift of 4 49 for the vinylic proton in cdcl3.
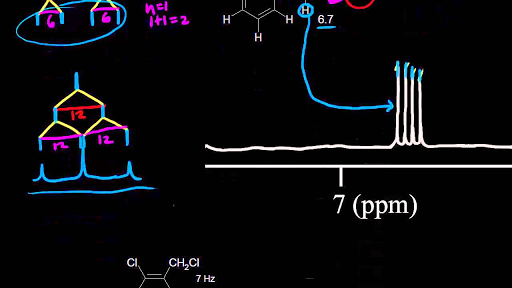
Chemical shift is associated with the larmor frequency of a nuclear spin to its chemical environment. This is a general trend add approximately 0 2 0 4 ppm for each additional alkyl group. C h acetylenic 2 3 ar h aromatic 6 8 5 ar c h benzylic 2 2 3 c c c h. Notice that the proton closest to the carbonyl group is at a higher chemical shift than the proton in cyclohexene 6 05 ppm for cyclohexenone vs.
Typical h nmr shift ranges. Chapters devoted to those groups.